Coisne C, Lyck R, Engelhardt B Live cell imaging techniques to study T cell trafficking across the blood-brain barrier in vitro and in vivo Fluids Barriers CNS. 2013 ;10(1):7. [Epub ahead of print]
BACKGROUND: The central nervous system (CNS) is an immunologically
privileged site to which access for circulating immune cells is tightly
controlled by the endothelial blood--brain barrier (BBB) located in CNS
microvessels. Under physiological conditions immune cell migration
across the BBB is low. However, in neuroinflammatory diseases such as multiple sclerosis,
many immune cells can cross the BBB and cause neurological symptoms.
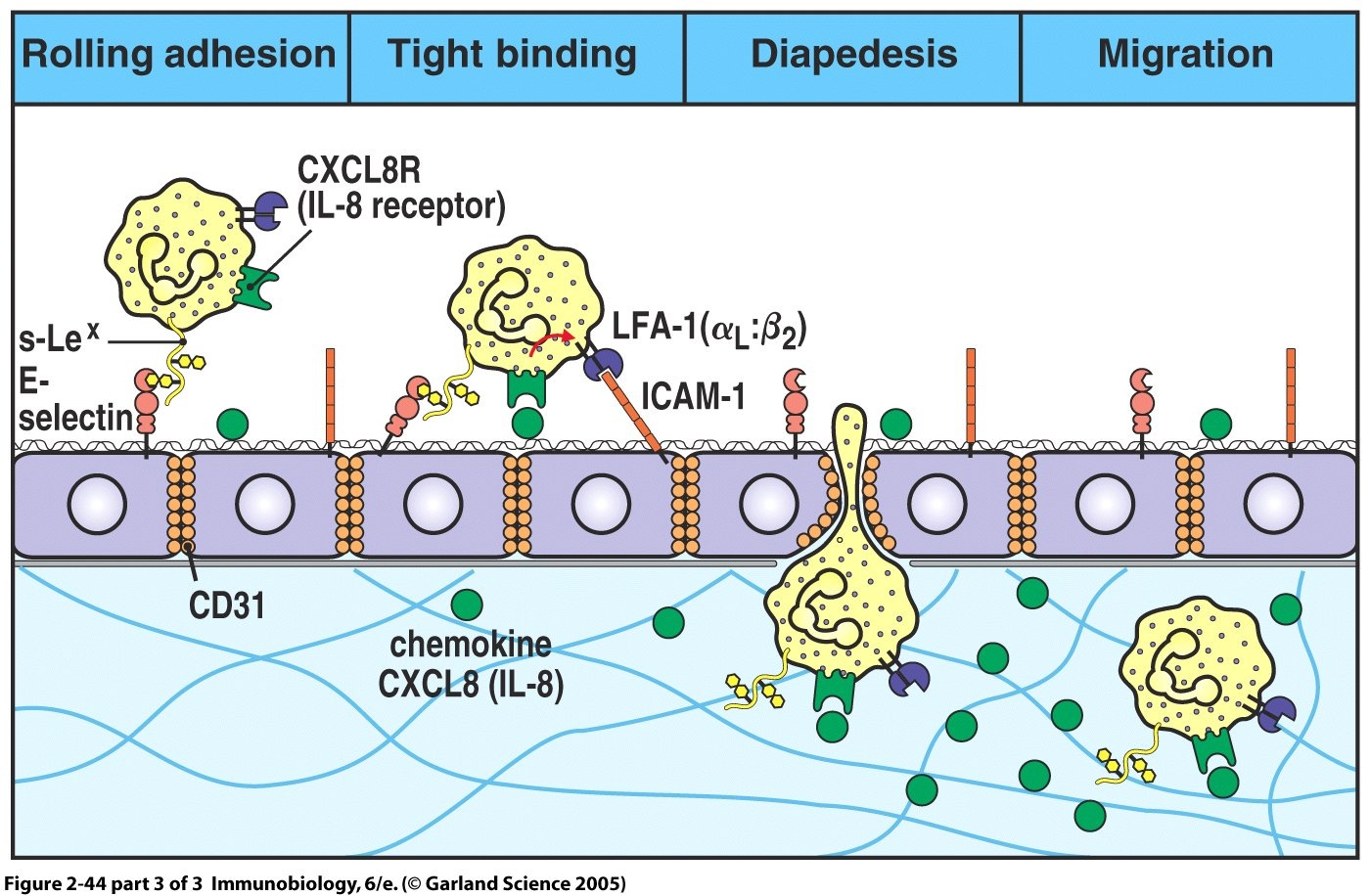
Extravasation of circulating immune cells is a multi-step process that
is regulated by the sequential interaction of different adhesion and
signaling molecules on the immune cells and on the endothelium. The
specialized barrier characteristics of the BBB, therefore, imply the
existence of unique mechanisms for immune cell migration across the
BBB.Methods and design: An in vitro mouse BBB model maintaining
physiological barrier characteristics in a flow chamber and combined
with high magnification live cell imaging, has been established. This
model enables the molecular mechanisms involved in the multi-step
extravasation of T cells across the in vitro BBB, to be defined with
high-throughput analyses. Subsequently these mechanisms have been
verified in vivo using a limited number of experimental animals and a
spinal cord window surgical technique. The window enables live
observation of the dynamic interaction between T cells and spinal cord
microvessels under physiological and pathological conditions using real
time epifluorescence intravital imaging. These in vitro and in vivo live
cell imaging methods have shown that the BBB endothelium possesses
unique and specialized mechanisms involved in the multi-step T cell
migration across this endothelial barrier under physiological flow. The
initial T cell interaction with the endothelium is either mediated by T
cell capture or by T cell rolling. Arrest follows, and then T cells
polarize and especially CD4+ T cells crawl over long distances against
the direction of flow to find the rare sites permissive for diapedesis
through the endothelium. DISCUSSION: The sequential use of in vitro and
in vivo live cell imaging of T cells interacting with the BBB allows us
to delineate the kinetics and molecular determinants involved in
multistep extravasation of encephalitogenic T cells across the BBB.
Seeing how cells get into the brain can now be achieved using imaging in the living and tells us so much more information. It can lead to new treatments however there is already tysabri that hits this pathway. Are there others that are better, yes I bet there are, but we still have drawing were the cells are going through the junctions, but they actually go through cells and not the usually the junction..